This article by Dr. V. Azbel, an Independent consultant on tantalum capacitors investigates influence of the size of sintered tantalum pellets on stability during the tantalum oxide film formation.
This study examines the impact of geometric dimensions on the mechanical properties and stability of sintered porous tantalum pellets during oxide film formation.
All technological parameters, including powder grade, pressing density, sintering temperature and time, shrinkage, and oxygen content, were kept constant, with the sole variable being pellet size. The findings indicate that yield strength and heat dissipation, both influenced by neck size, are critical factors in determining stability during formation.
Introduction
Sintered porous tantalum pellets, produced from 20 kCV/g powder with a green density of 5.5 g/cm³ and sintered at 1620°C for 20 minutes, are utilized in the manufacturing of tantalum capacitor anodes. Given identical sintering conditions, shrinkage was uniform across samples.
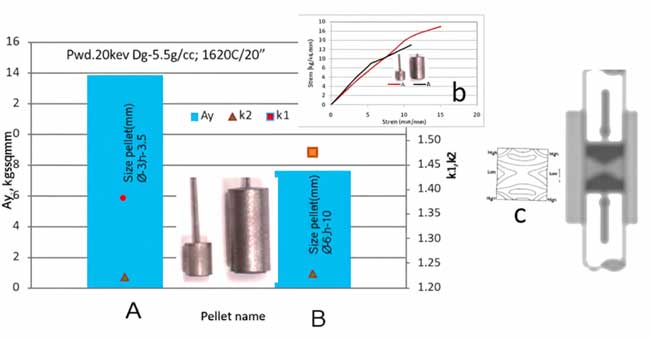
The morphology of the porous structure significantly affects the pellets’ behavior during oxide film formation. Stress-strain curve characteristics were employed as numerical indicators. Previous research has shown that yield strength, Young’s modulus (k₁), and the hardening coefficient (k₂) are contingent upon pressing and sintering conditions. In this study, all parameters and pellet shapes were consistent, allowing for an exploration of the effects of pellet size on thermal conductivity, internal stresses, and stability during formation.
Experimental
In an experimental study, tantalum powder of uniform grade, with a specific gravity of 20 kCV/g, was used to create pellets under identical processing conditions, including temperature, time, density, shrinkage, and oxygen content.
The pellets were categorized into two sizes: Pellet A, which had a smaller diameter and height, and Pellet B, which was larger in dimensions. After sintering, both pellets were subjected to electrolytic formation at the same voltages using two different electrolytes: 0.01% H₃PO₄ and a mixture of 50% glycol with 0.05% H₃PO₄. These electrolytes differ in conductivity, which affects the electrolysis rate and heat dissipation, with the formation current normalized to mass in amperes per gram.
Processing Conditions: Pellets A and B were processed under identical conditions (temperature, time, density, shrinkage, oxygen content).
Samples:
- Pellet A: Smaller diameter and height.
- Pellet B: Larger dimensions.
| Pellet Size | Pellet A | Pellet B |
|---|---|---|
| diameter [mm] | 3.1 | 4.83 |
| height [mm] | 1 | 10 |
Post-sintering, the pellets underwent electrolytic formation at identical voltages in two electrolytes:
- 0.01% H₃PO₄
- 50% glycol + 0.05% H₃PO₄
These electrolytes vary in conductivity, influencing the electrolysis rate and heat dissipation. The formation current was normalized to mass (A/g).
Results
The results, as depicted in Figure 1, include combined data on mechanical parameters, stress-strain diagrams, and stress distribution under compression for both pellets.
Pellet A demonstrated a yield strength approximately 40% higher than Pellet B, suggesting a larger cross-section of necks in its porous structure and superior heat dissipation. The variation in pellet size led to different stress distributions and density distributions, influencing neck size.
During the formation process, in the 0.01% H₃PO₄ electrolyte, Pellet A maintained its integrity, while Pellet B was destroyed. However, in the low-conductivity electrolyte consisting of 50% glycol and 0.05% H₃PO₄, both pellets underwent formation without destruction. The reduced conductivity in this electrolyte slowed down film growth and heat release, ensuring stability even for pellets with lower yield strength.
In summary – behavior during formation:
- In 0.01% H₃PO₄ Electrolyte:
- Pellet A maintains integrity.
- Pellet B is destroyed.
- In Low-Conductivity Electrolyte (50% glycol + 0.05% H₃PO₄):
- Both pellets undergo formation without destruction.
- Reduced conductivity slows film growth and heat release, ensuring stability even for pellets with lower yield strength.
Discussion
The discussion highlights several key points:
1. Heat Dissipation and Yield Strength Yield strength is correlated with the cross-sectional size of necks in the porous structure. Higher yield strength, at similar k₂ values, enhances heat dissipation.
2. Role of Pellet Size: Differences in pellet dimensions lead to varied stress distributions and heat dissipation capabilities.
3. Electrolyte and Film Growth Rate**: Lower electrolyte conductivity reduces oxide film growth rate, heat generation, and enhances pellet stability during formation.
4. Indirect Estimation of Stress Distribution: Stress distributions are not directly observable but can be inferred from stress-strain curves.
Conclusion
In conclusion, under identical technological parameters, the behavior of sintered tantalum pellets during formation is solely influenced by their size. Pressing and sintered density are not reliable indicators of the porous structure. Yield strength, correlating with neck size and heat dissipation, is the key factor in stability. For pellets with lower yield strength, reducing the oxide film growth rate using electrolytes of lower conductivity decreases heat generation and prevents destruction.
Appendix
Strain-Stress Curve Parameters for Porous Materials
Yield Stress (Ay): The yield stress of porous materials is related to the ratio of neck size (X) to primary powder particle size (D) by the equation:
Ay = b*A0*(X/D)², where b is an empirical constant, Ay is the yield point of the sintered porous material, and A0 is the yield point of the deformed material.
Young’s Modulus (k1 ~ E): Described by the equation: k1 ~ E = E0 (1-p/pc), where E is the elastic modulus of a porous material, E0 is the modulus of solid material at density pc ~ 1, and p is the porosity-related density.
Hardening Coefficient (k2 ~ P): An exponential coefficient correlating with material defectiveness.
Note: The size D determines BET.
References
Vladimir Azbel: Structural Interpretation of Reliability in Tantalum Capacitor Anodes