Researchers from China developed an in‑situ template–activation route using sodium lignosulfonate and potassium oxalate to synthesize 2D porous carbon cathode nanosheets optimized for zinc‑ion hybrid supercapacitors.
This scalable, environmentally friendly method yields a high‑surface‑area LSC‑800 cathode that delivers high capacitance, high energy density, and excellent long‑term cycling stability in aqueous ZIHCs.
Introduction
Two-dimensional porous carbon materials have emerged as highly promising cathodes for aqueous zinc‑ion hybrid supercapacitors, combining large accessible surface area with abundant electrochemically active sites. However, most existing synthesis routes rely on corrosive activators, external templates, or multi‑step procedures that are difficult to scale and environmentally problematic. This article addresses the need for a simple, green, and scalable strategy to produce 2D porous carbons with optimized pore architecture tailored for hydrated zinc ion storage.
The authors propose an in‑situ template–activation approach based on co‑pyrolysis of a homogeneous composite of low‑cost sodium lignosulfonate and potassium oxalate. During heat treatment, potassium oxalate decomposes to potassium carbonate, which simultaneously templates the 2D structure and activates the carbon framework to generate a hierarchical, micropore‑rich network. The resulting 2D porous carbons, denoted LSC‑T, are evaluated as cathode materials for zinc‑ion hybrid capacitors, demonstrating high capacity, energy density, and outstanding cycling stability.
Key points
- The work introduces an in‑situ template–activation strategy using potassium oxalate as both structural template and chemical activator for 2D porous carbons derived from sodium lignosulfonate.
- Potassium oxalate decomposes into potassium carbonate during pyrolysis, which acts as a core to direct formation of interlaced 2D nanosheets and to etch nanopores in the outer carbon layer.
- The method avoids external templates, strongly corrosive activators such as KOH/NaOH, and post‑treatments with strong acids or bases, making the route more environmentally benign and operationally simple.
- The resulting LSC‑T carbons exhibit high specific surface areas, hierarchical porosity, and abundant surface functional groups, with a uniform micropore size distribution.
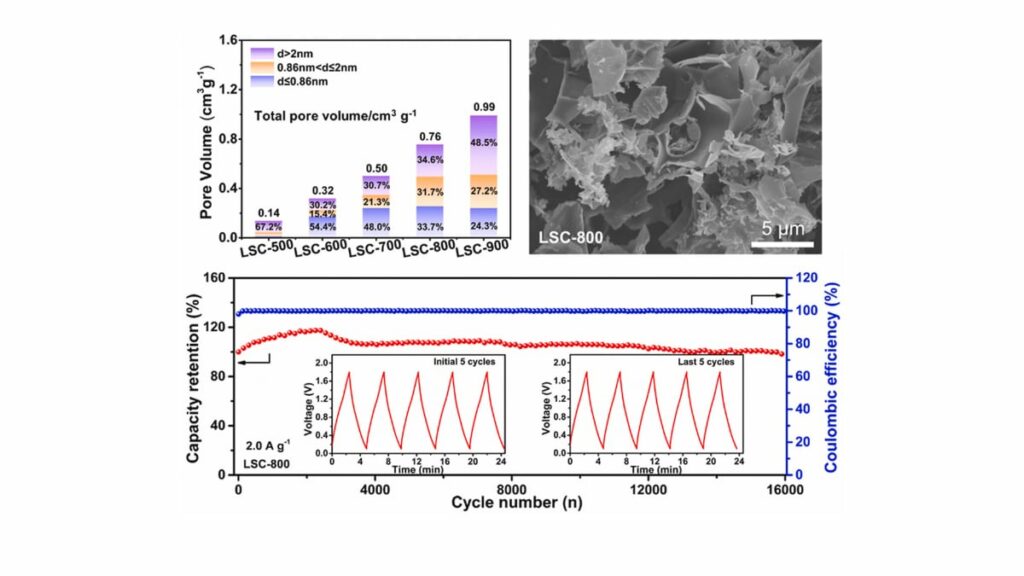
- The optimized sample LSC‑800 achieves a specific surface area of 1546.3 m² g−1^{-1}−1 and a micropore volume ratio of 31.7% in the 0.86–2.0 nm range, accompanied by additional mesoporosity.
- When used as the cathode in zinc‑ion hybrid supercapacitors, LSC‑800 delivers a high specific capacity of 172.2 mAh g−1^{-1}−1 and an energy density of 152.9 Wh kg−1^{-1}−1 at 0.1 A g−1^{-1}−1.
- The assembled zinc‑ion hybrid supercapacitors exhibit long‑term cycling stability of 16 000 cycles at 2.0 A g−1^{-1}−1 with about 98% capacity retention.
- The zinc storage mechanism involves not only electric double‑layer behavior but also reversible formation and dissolution of a basic zinc sulfate phase Zn4_44SO4_44(OH)6_66·5H2_22O on the cathode surface.
- The study demonstrates that combining 2D morphology, tailored micropore–mesopore architecture, and abundant surface sites can significantly enhance Zn2+^{2+}2+ storage in hybrid capacitor configurations.
- The proposed route is presented as an efficient, feasible platform for designing high‑performance 2D porous carbon cathodes for zinc‑ion hybrid supercapacitors and potentially other energy storage devices.
Extended summary
Aqueous zinc‑ion hybrid capacitors (ZIHCs) combine a battery‑type zinc metal anode with a capacitor‑type carbon cathode, aiming to synergistically merge high energy density with high power density and safety. Conventional lithium‑ion batteries suffer from safety concerns related to flammable organic electrolytes, whereas aqueous zinc systems benefit from low cost, environmental compatibility, and inherently safer electrolytes. The zinc anode offers high theoretical capacity and low redox potential, but the performance of ZIHCs is often limited by the porous carbon cathode. In particular, typical carbon cathodes operating purely via electric double‑layer charge storage show relatively low capacity, and their pore networks are frequently sub‑optimal for the large hydrated zinc complexes that dominate in aqueous electrolytes.
In aqueous media, zinc ions are primarily present as hydrated complexes, often represented as [Zn(H2O)6]2+\text{[Zn(H}_2\text{O)}_6]^{2+}[Zn(H2O)6]2+, with a large effective hydration diameter. When carbon electrodes possess narrow micropores that are not matched to this hydration size, diffusion becomes sluggish and only a fraction of the pore volume is effectively used. Many existing activated carbons have high overall specific surface areas but pore size distributions heavily skewed toward very small micropores, which are not easily accessible to hydrated zinc species. Moreover, conventional activation routes typically employ strong alkali activators such as KOH or NaOH, generating corrosive waste and requiring extensive washing, and they often produce three‑dimensional bulk carbons with poorly connected pores and non‑uniform pore distributions.
Two‑dimensional porous carbons, particularly in the form of nanosheets, offer advantages in ionic transport and electrode–electrolyte contact. Reduced thickness shortens diffusion paths and can expose more active sites on the surface, while hierarchical porosity can facilitate rapid ion transport and charge storage. Previous studies have used sacrificial templates such as ZnO, CaO, or NaCl to form 2D carbon nanosheets, but these approaches typically involve multiple steps, including template synthesis or mixing, high‑temperature treatment, and subsequent template removal with acids or other reagents. Furthermore, they tend to favor meso‑ and macroporosity with relatively low specific surface areas, which may not fully leverage the zinc‑ion storage potential of a 2D architecture.
The present work addresses these challenges by designing an in‑situ template–activation strategy. Sodium lignosulfonate, a lignin‑based biomass by‑product, is chosen as a cheap and abundant carbon precursor. Potassium oxalate (K2_22C2_22O4_44) is introduced as a green, mild, and low‑cost activator, building on prior work that used this salt to activate biomass carbons in a single pyrolysis step. The authors dissolve sodium lignosulfonate and potassium oxalate in water to form a homogeneous solution, then evaporate the solvent to obtain a uniform solid composite. Upon pyrolysis at different temperatures (denoted by LSC‑T, where T indicates the temperature), potassium oxalate decomposes to potassium carbonate (K2_22CO3_33), which plays a dual role.
First, the in‑situ generated potassium carbonate acts as a template core that promotes development of a 2D, interlaced nanosheet morphology. The intimate mixing of the carbon precursor and K2_22C2_22O4_44 at the molecular or nanoscale ensures that upon decomposition, the evolving K2_22CO3_33 is uniformly embedded within the carbon matrix. This leads to a self‑templating effect where gas evolution and phase transformations help shape the carbon into thin sheets that intersect and interlace, creating a large accessible surface. Second, K2_22CO3_33 serves as an activator that reacts with the surrounding carbon at elevated temperature, etching nanopores and generating a hierarchical pore structure dominated by micropores with supplementary mesopores. Because the activator is generated in situ and consumed during activation, there is no need to add external templates or corrosive hydroxides, nor to perform aggressive post‑treatments with strong acids or bases to remove solid residues.
The authors perform morphological and structural characterization of the LSC‑T series obtained at different calcination temperatures. The synthesis scheme, depicted in their Scheme 1, highlights the steps of dissolution, drying, and thermal treatment leading to 2D porous carbons. The characterization confirms that the materials consist of two‑dimensional, interlaced nanosheets with well‑developed porosity. Nitrogen adsorption–desorption measurements reveal that the carbons possess high specific surface areas and hierarchical pore size distributions. Critically, the optimized sample LSC‑800, produced at 800 °C, exhibits a specific surface area of 1546.3 m² g−1^{-1}−1. The micropore volume fraction is reported as 31.7%, with pore sizes concentrated in the range of 0.86–2.0 nm, which is considered favorable for accommodating hydrated zinc species while still providing strong confinement and high surface area. Additional mesoporosity is present, which helps facilitate electrolyte penetration and rapid ion transport to and from the microporous regions. The materials also feature abundant surface functional groups, which may contribute to wettability and pseudocapacitive interactions with zinc ions.
Electrochemical evaluation is carried out by assembling zinc‑ion hybrid capacitors using zinc metal as the anode and LSC‑T materials as the cathodes. The devices are operated in an aqueous electrolyte environment suitable for Zn2+^{2+}2+ transport. The LSC‑800 cathode delivers a specific capacity of 172.2 mAh g−1^{-1}−1 at a current density of 0.1 A g−1^{-1}−1, which represents a notably high value for carbon‑based ZIHC cathodes. The corresponding energy density reaches 152.9 Wh kg−1^{-1}−1 at the same current density, demonstrating that the combination of high surface area, tuned microporosity, and 2D architecture translates into excellent charge storage performance. At higher current densities, the devices maintain competitive capacity and power characteristics, although detailed rate performance parameters are not reproduced here to avoid reconstructing the article.
Cycling stability is a crucial metric for practical hybrid capacitors. The assembled ZIHCs with LSC‑800 cathodes are reported to endure 16 000 charge–discharge cycles at 2.0 A g−1^{-1}−1 with an approximate capacity retention of 98%. This indicates minimal degradation and suggests that the carbon framework and zinc‑ion storage mechanism are highly reversible under the tested conditions. Structural robustness of the 2D network and stable electrode–electrolyte interfaces contribute to preserving performance over extended operation.
Beyond global performance metrics, the authors discuss the zinc‑ion storage mechanism in these 2D porous carbons. While electric double‑layer capacitance arising from ion adsorption at the carbon/electrolyte interface remains a core component, additional contributions from surface or near‑surface redox processes are indicated. The formation and dissolution of a basic zinc sulfate phase, Zn4_44SO4_44(OH)6_66·5H2_22O, on the cathode surface are observed and linked to the storage mechanism. This phase likely forms during charging–discharging due to local supersaturation and interactions between Zn2+^{2+}2+, sulfate anions in the electrolyte, and hydroxide generated by electrochemical processes. Its reversible evolution suggests a hybrid charge storage behavior combining capacitive adsorption and fast surface‑limited conversion reactions, which can help explain the high capacity and good rate response.
By integrating materials chemistry, pore‑structure engineering, and electrochemical analysis, the study establishes a clear relationship between synthesis strategy, microstructure, and device performance. The in‑situ template–activation route enables fine control over pore size distribution and 2D morphology without resorting to environmentally harmful reagents or complicated multi‑step protocols. The LSC‑800 sample exemplifies how carefully tuned micropores in the sub‑2 nm range, together with supporting mesopores and a thin‑sheet architecture, can accommodate large hydrated zinc complexes and provide rapid ion transport pathways, leading to improved energy and power characteristics in ZIHCs.
Conclusion
The article presents an in‑situ template–activation strategy for fabricating 2D porous carbon nanosheets from a sodium lignosulfonate and potassium oxalate precursor, achieving high specific surface area, tailored microporosity, and interlaced sheet morphology in a single, environmentally benign process. By leveraging potassium oxalate as both template precursor and activator, the method avoids corrosive activators and complex template removal, yet yields carbons that perform exceptionally well as cathodes in zinc‑ion hybrid supercapacitors. The optimized LSC‑800 material supports high specific capacity and energy density, along with excellent long‑term cycling stability, highlighting the importance of pore size matching and 2D architectures for zinc‑ion storage.
Limitations include the focus on a specific biomass and activator system, and the absence in the abstract‑level text of detailed mechanistic quantification of individual charge storage contributions. Future work could expand the approach to other biomass sources and heteroatom dopants, refine pore architecture for even higher rate capability, and deepen understanding of the interplay between electric double‑layer behavior and basic zinc sulfate phase formation in these hybrid capacitors.
References
- Shanxing Wang, Renjie Zhao, Rongzhi Peng, Zhu Liu, Feng Xiao, Yubo Zou, Guocong Liu, “An in‑situ template‑activation strategy to construct 2D porous carbon with interlaced nanosheets for zinc‑ion hybrid capacitors,” Carbon, Volume 247, February 2026, Article 121068. Available at: https://www.sciencedirect.com/science/article/abs/pii/S000862232501084X